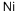化学品概述
化学品概述
 基本信息
基本信息
- 中文名称:
- 中文别名:
- 英文名称:
- 英文别名:
- 分子式:
- 分子量:
- CAS:
- EINECS编号:
- MDL编号:
- 精确质量:
- InChI:
- InChI Key:
- MOL文件:
- PSA:
- LogP:
- FEMA编码:
- COE编码:
 理化性质
理化性质
-
外观性质:
wire;White to gray-white
-
熔点:
212 °C (dec.)(lit.)
-
沸点:
2732 °C(lit.)
-
密度:
8.9
- 折射率:
- 比旋光度:
- 闪点:
- 溶解性:
-
酸度系数(pKa):
It is insoluble in water.
- 相对极性:
-
PH值:
8.5-12.0
- 爆炸极限值(explosive limit):
-
敏感性:
air sensitive
-
储存条件:
Flammables area
- 检测方法:
- 蒸气压:
-
Merck:
14,8107
- BRN:
-
NIST化学物质信息:
http://webbook.nist.gov/cgi/cbook.cgi?ID=7440-02-0&Units=SI
-
EPA化学物质信息:
http://iaspub.epa.gov/sor_internet/registry/substreg/searchandretrieve/advancedsearch/search.do?search=?search=&searchCriteria(advancedCriteria)=casNumber=7440-02-0
 安全信息
安全信息
-
危化品标志:
C,Xi,Xn,F,T
-
危化代码:
34-50/53-43-40-10-17-52/53-48/23
-
安全代码:
26-45-60-61-36-22-36/37-16-15-5-36/37/39-43-28
- 海关编码/HS编码:
-
危化品运输编码:
UN 1493 5.1/PG 2
-
WGK Germany:
3
-
RTECS:
VW4725000
-
TSCA:
Yes
-
危化等级:
4.1
-
包装类别:
II
- 毒理资料:
- 灭火剂:
 应用领域
应用领域
主要用于制造电碳制品、摩擦材料、含油轴承及粉末冶金结构材料用作加氢催化剂,也用于镍盐制造用于生产优质不锈钢、高温高强度合金、精密合金、其他含镍合金、加工纯镍、电真空用镍等镍制品用作催化剂分析试剂.用用于金刚石刀头,锯片,磨轮,空心薄壁钻等产品行业。用于活性物质的导电添加剂,其独特的链珠状结构可产生良好的导电网络,应用于制作导电涂料、油漆、胶粘剂,导电胶带等行业。油类和其他有机物质氢化反应的催化剂,制备镍盐,电镀,合金。
 制备方法/合成路线
制备方法/合成路线
参考资料
- Ni; at. wt 58.6934; at. no. 28; valence 2; seldom 1, 3, 4. Group VIII (10). Naturally occurring isotopes (mass numbers): 58 (68.27%), 60 (26.10%), 61 (1.13%), 62 (3.59%), 64 (0.91%); known artificial radioactive isotopes: 53, 55-57, 59 (longest-lived known isotope, T½ 7.5 ´ 104 years, decay by electron capture), 63, 65-67. Abundance in earth's crust 99 ppm. Discovered by Cronstedt in 1754: Cronstedt, Mineralogie (Stockholm, 1758) p 218. Isoln: Berthier, Ann. Chim. Phys. [2] 14, 52 (1820); 25, 94 (1824). Occurs free in meteorites. Found in many ores as sulfides, arsenides, antimonides and oxides or silicates; chief sources include chalcopyrite, q.v., pyrrhotite, pentlandite [(Fe,Ni)9S8] and garnierite [3(Mg,Ni)O.2SiO2.2H2O]; other ores include niccolite (NiAs) and millerite (NiS). Methods of extraction and purification: Mackiw, Can. J. Chem. Eng. 46, 3 (1968); Houot, Ann. Mines 1969 (April), 9; Queneau, J. Met. 22, 44-48 (1970). Prepn of high purity nickel: Wise, Schaefer, Metals Alloys 16, 424 (1924); from NiO and H2: Glemser in Handbook of Preparative Inorganic Chemistry vol. 2, G. Brauer, Ed. (Academic Press, New York, 2nd ed., 1965) pp 1543-1544; by electrolysis: Vu Quang Kinh, Nardin, C.R. Seances Acad. Sci. Ser. C 266, 307 (1968). Comprehensive reviews: Gmelins, Nickel (8th ed.) 57, 5 vols, about 3500 pp (1965-1967); Nicholls in Comprehensive Inorganic Chemistry vol. 3, J. C. Bailar, Jr. et al., Eds. (Pergamon Press, Oxford, 1973) pp 1109-1161; J. K. Tien, T. E. Howson in Kirk-Othmer Encyclopedia of Chemical Technology vol. 15 (Wiley-Interscience, New York, 3rd ed., 1981) pp 787-801; Chemistry of the Elements N. N. Greenwood, A. Earnshaw, Eds. (Pergamon Press, New York, 1984) pp 1328-1363. Book: Nickel Toxicology, S. S. Brown, F. W. Sunderman, Eds. (Academic Press, New York, 1980) 193 pp. Review of carcinogenic risk: IARC Monographs 11, 75-112 (1976); of toxicology and human exposure: Toxicological Profile for Nickel (PB2006-100005, 2005
 MSDS
MSDS
 MSDS
MSDS
 图谱
图谱
 计算化学数据
计算化学数据
- 疏水参数计算参考值(XlogP):
-
氢键供体数量 :
0
-
氢键受体数量:
0
-
可旋转化学键数量:
0
-
拓扑分子机型表面积(TPSA) :
0
-
重原子数量:
1
-
形式电荷:
0
-
复杂度:
0
-
同位素原子数量:
0
-
确定原子立构中心数量:
0
-
不确定原子立构中心数量:
0
-
确定化学键立构中心数量:
0
-
不确定化学键立构中心数量:
0
-
共价键单元数量:
1